
Kabrita can reduce GI discomfort
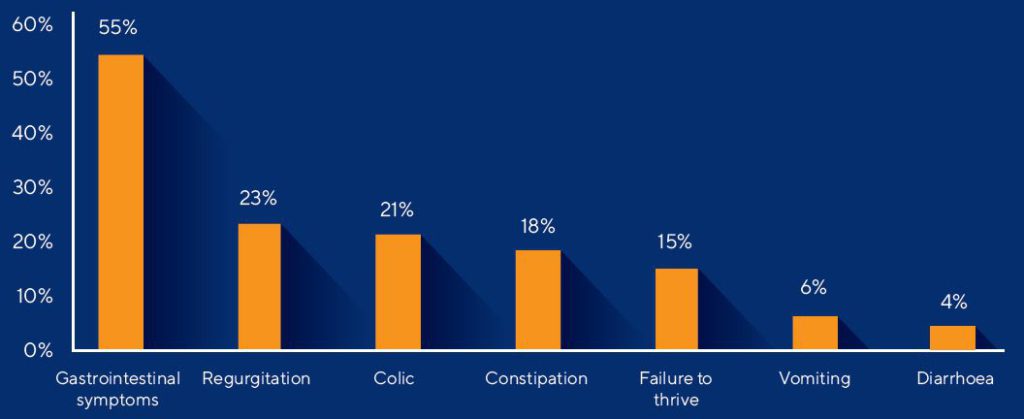
The early phase of life is a period of rapid growth and development. Minor digestive disturbances in both breastfed and formula-fed infants are often reported. These disturbances could be due to the maturation of the digestive and immune system as well as the development of the gut microbiota. Gastrointestinal symptoms were seen in 54% of infants in a population-based prospective study.1 These infants were (partially) breastfed and formula-fed. The most frequently reported symptoms of discomfort were hiccups, crying, bloating, flatulence and difficulties related to stools (hard and soft stools) (see figure below).1,2,3 Some of these minor disturbances could become more severe and could be diagnosed as functional gastrointestinal disorders according to the Rome IV criteria.
Digestive disturbances in the infant can lead to parental concerns.4 Parents can experience stress and suffer from lack of sleep.4,5,6 Although the infants may experience minor disturbances, most of the infants are otherwise healthy and are not diagnosed for functional gastrointestinal disorders and do not need any medical treatment.4 Healthcare professionals can differentiate between functional gastrointestinal disorders and minor discomfort that the infants will naturally outgrow. Many parents may switch nutrition in their search for relief of their infant’s digestive disturbances. Switching formula can be stressful and could result in lower volume intake and should preferably be prevented (data on file).
An infant formula that is easier to digest may prevent or resolve digestive discomfort and the need to switch to another infant formula. According to parents, gentle infant formula is easily digestible and is associated with relaxed feeding moments and more joyful moments with the infant (based on US market research among parents).
Kabrita can reduce gastro-intestinal discomfort: a randomized controlled trial
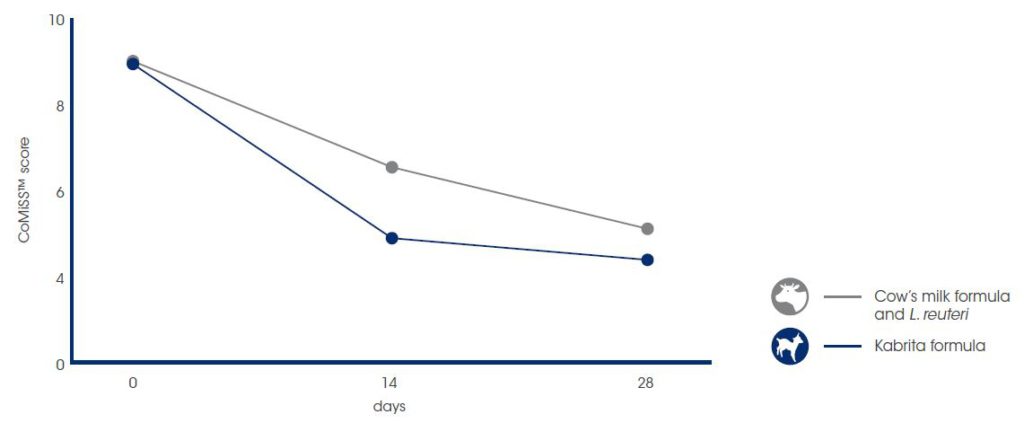
Ausnutria’s research team started to investigate the beneficial effects of Kabrita formula on comfort in infants with mild gastro-intestinal (GI) issues.7 In this randomized controlled study, 9 infants used Kabrita formula for 28 days, while a control group of 7 infants used a CMF with probiotics supplements. The use of a CMF in combination with Lactobacillus reuteri is the standard medical treatment for infants with mild GI complaints in Spain.8 All infants were aged between 1 and 3 months and had mild GI discomfort based on a total CoMiSSTM score between 6 and 12. CoMiSSTM (Cow’s Milk-related Symptom Score) is a scoring tool ranging from 0-33 that sums up the GI-related complaints in 5 different categories, namely crying, stools, reflux, skin and respiratory issues. A CoMiSSTM score of 0 indicates ‘no discomfort’, while a CoMiSSTM score between 6 and 12 indicates ‘mild discomfort. After two weeks intervention, the GI discomfort significantly decreased in both groups. However, the GI discomfort decreased faster in the Kabrita group compared to the control group after 14 days of intervention. The decrease in GI discomfort was mainly due to decreased crying and improved stool consistency.
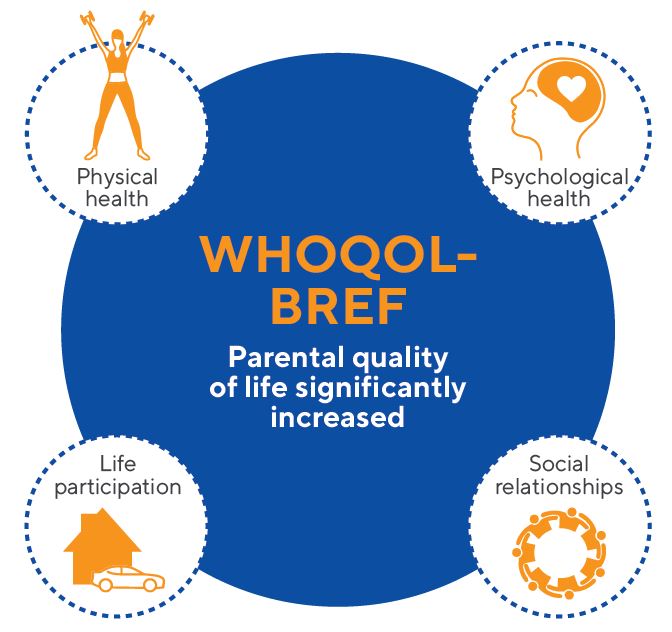
Parental quality of life was assessed with the WHOQOL-BREF, which is a questionnaire developed by the World Health Organization that assesses the quality of life based on four indicators (see figure below). Parental quality of life significantly improved in the Kabrita group after two weeks. However, quality of life of parents in the Kabrita group was comparable to that of the CMF group.
Based on this study, it can be concluded that Kabrita formula can decrease GI discomfort faster (nonsignificant trend) in infants with mild GI issues as compared to CMF.7
Click on the poster below for more details about this study.

Kabrita real-world evidence data: improvement in infants’ comfort
Two real-world evidence studies show that infants consuming Kabrita have significantly fewer GI symptoms and skin symptoms than infants not consuming GMF, based on a lower CoMiSS® score.9,10 These results indicate that infants can benefit from GMF in managing GI symptoms and skin symptoms.
Kabrita and the prevalence of GI symptoms in infants around the world
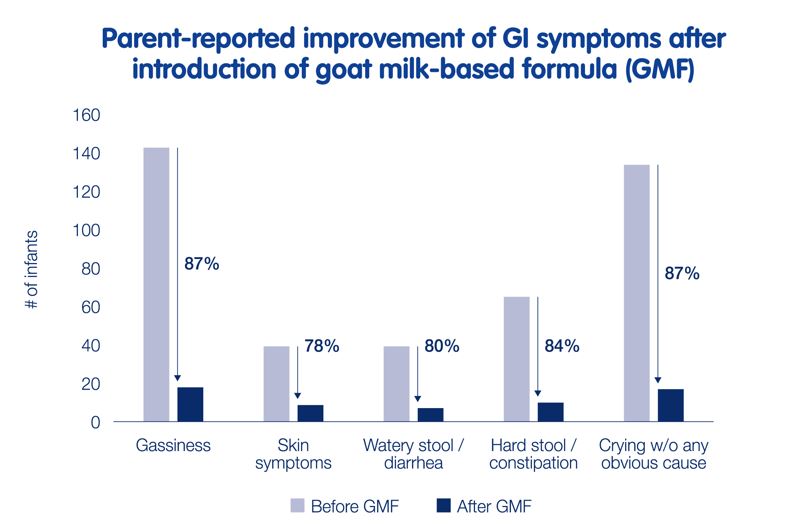
An improvement in GI comfort was also seen in a real-world evidence study amongst Kabrita consumers in Brazil, Mexico, Russia and the Netherlands.9 Parents of infants using Kabrita filled out 595 questionnaires, of which 425 replies met the inclusion criteria for infants within 0-24 months old and no medication use. Parents reported improvement of symptoms after introduction of the goat milk-based formula Kabrita: less gassiness, fewer skin symptoms, less frequent watery stools/diarrhoea, less frequent hard stools/constipation and less crying without any obvious cause.
In the group of infants consuming Kabrita, the CoMiSS® was low (median 1.0), and 89% had a CoMiSS® ≤ 6, indicating that the infants were healthy. The total CoMiSS® was significantly lower (meaning fewer symptoms) for Kabrita infants as compared to non-goat milk-formula infants (p=<0.001). More details about this study can be found on the poster presented at ESPGHAN 2023 below.

ESPGHAN poster real-world evidence
Fewer gastrointestinal symptoms in infants consuming Kabrita
Another real-world evidence study in Chinese infants (n=767) studied the effect of Kabrita (n=303) and CMF (n=464) on GI symptoms in infants by using online questionnaires.10 The results showed that the overall CoMiSS® (median 2) was low, indicating the infants were healthy. However, the total CoMiSS® was significantly lower for the infants consuming Kabrita compared to the infants consuming CMF (p<0.001), mainly due to fewer regurgitation and skin symptoms for the Kabrita consumers.
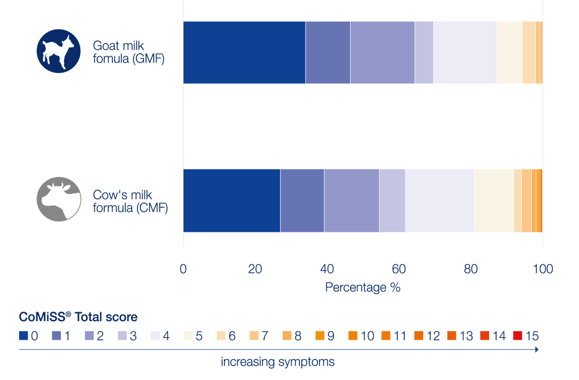
More details about this study can be found on the poster presented at ESPGHAN 2023 below.

ESPGHAN poster Chinese infants
Other studies confirm: goat milk formula has positive effect on infants’ comfort
The positive effects of goat milk formula on comfort in infants were also shown in other studies testing goat milk formulas. One study investigated the effects of a goat milk formula in infants with constipation, aged between 1 and 4 months of age.11 The enrolled infants were ill and visited a paediatric clinic for treatment of their constipation. Twenty infants completed the study and had softer stools after three weeks of GMF. In addition, the number of hours that infants cried reduced significantly during the period in which they took GMF from 3 hours to 1 hour per 24 hours. Parents reported a change in infant behaviour within three to five days after starting with the GMF. This indicates that a goat milk-based formula can improve GI comfort, although well designed studies are warranted to confirm these findings.
HCP survey: Cow’s Milk Disorders and the Role of Goat Milk-Based Formulas
In collaboration with the EAP (Egyptian Acadamy of Pediatrics), Ausnutria’s R&D team investigated the understanding of physicians regarding Cow’s milk Allergy (CMA) and Cow’s Milk Intolerance (CMI) in the Middle East and North Africa region.12 Together with an expert panel, supervised by Prof. Vandenplas, the questionnaire was developed. The survey was send out to 2500 health care professionals practising in the region. In addition, the potential role of goat milk formula was also explored as an alternative nutrition for infants suffering from CMI was.
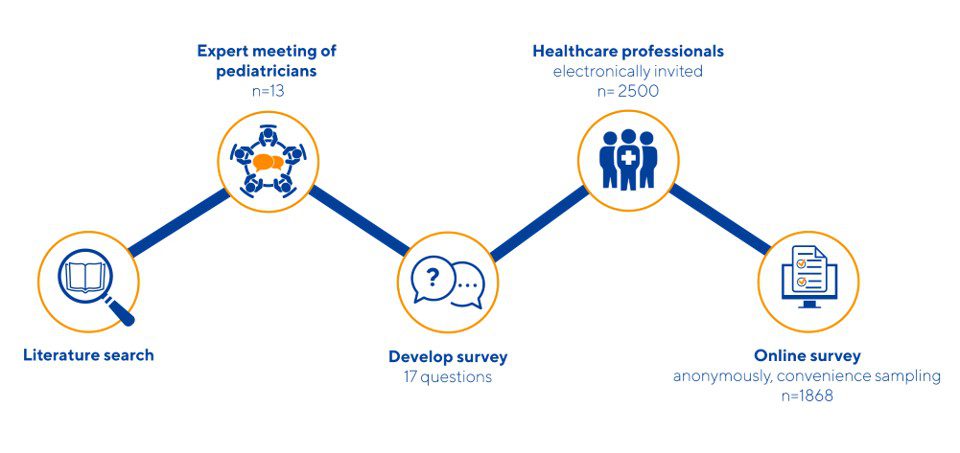
The paper concluded that doctors in general are aware of the difference between CMA and CMI but practice seems to be more complex. Next to this, goat milk formula was evaluated as a promising alternative for infants with CMI, due to it’s beneficial nutritional profile as 72% of the doctors mentioned a better protein digestibility of goat milk formula compared to cow’s milk.
Click on the paper below for more information about this study.

Paper: HCP consensus CMI
Please note: goat milk formula is not a suitable alternative in case of a medically diagnosed CMA, due to possible cross-reactivity.
Conclusion
Kabrita formula reduced GI discomfort faster than cow’s milk formula in infants with mild GI complaints and can be of value for infants with GI symptoms associated with cow’s milk due to cow’s milk intolerance.
References
- 1. Iacono, G., et al., Gastrointestinal symptoms in infancy: a population-based prospective study. Dig Liver Dis, 2005. 37(6): p. 432-8.
- 2. Neu, J., Gastrointestinal maturation and implications for infant feeding. Early Hum Dev, 2007. 83(12): p. 767-75.
- 3. Hamosh, M., Digestion in the newborn. Clin Perinatol, 1996. 23(2): p. 191-209.14
- 4. Vandenplas, Y., Algorithms for Common Gastrointestinal Disorders. J Pediatr Gastroenterol Nutr, 2016. 63 Suppl 1: p. S38-40.
- 5. Bellaiche, M., et al., Multiple functional gastrointestinal disorders are frequent in formula-fed infants and decrease their quality of life. Acta Paediatr, 2018. 107(7): p. 1276-1282.
- 6. Benninga, M.A., et al., Childhood Functional Gastrointestinal Disorders: Neonate/Toddler. Gastroenterology, 2016.
- 7. Meijer-Krommenhoek, Y., et al. Goat milk based infant formula improves gastro-intestinal discomfort in infants in a randomized controlled pilot study. Poster presented at Nutrition & Growth 2021.
- 8. Jiménez Ortega, A., et al., Nutrición y microbiota en población pediátrica. Implicaciones sanitarias. Nutr. Hosp. , 2020. 37
- 9. Böhme, J., et al. Goat milk-based infant formula and the prevalence of gastrointestinal symptoms in infants: A real world evidencestudy from Brazil, Mexico, Russia and The Netherlands. ESPGHAN 55th Annual Meeting Abstracts. Pediatric Gastroenterology and Nutrition 76(S1):p 1-1407, June 2023
- 10. Zhong, J., et al. Less gastrointestinal symptoms in infants consuming goat milk-based infant formula: a-real-world-evidence study. ESPGHAN 55th Annual Meeting Abstracts. Pediatric Gastroenterology and Nutrition 76(S1):p 1-1407, June 2023
- 11. Infante, D., G. Prosser, and R. Tormo, Constipated Patients Fed Goat Milk Protein Formula: A Case Series Study. Journal of Nutrition and Health Sciences, 2018. 5(2): p. 1-6.38
- 12. Bahbah W.A., et al. A Survey to Identify the Current Management of Cow’s Milk Disorders and the Role of Goat Milk-Based Formulas in the Middle East and North Africa Region. Nutrients. 2022 Mar 3;14(5):1067.



